Introduction
A polymer is a material composed of large molecules formed by the repeated connection of smaller structural units, known as monomers. These repeating units are chemically bonded to form long chains or branched networks, giving polymers distinctive physical and chemical properties such as high molecular weight, flexibility, and strong interaction with surrounding fluids. Because of these characteristics, polymers are widely used in industrial and engineering applications, including enhanced oil recovery, where they are employed to modify fluid flow behavior.
Xanthan gum is a natural polymer. Its structure consists of a backbone made of repeating sugar units, with side chains attached at regular intervals. This branched configuration allows xanthan gum molecules to strongly interact with water, forming a stable and viscous solution. The presence of multiple hydroxyl and charged groups along the polymer chain enhances its ability to bind water molecules, which explains its strong thickening behavior even at relatively low concentrations [10, p. 5].
Polyacrylamide is a synthetic polymer formed from acrylamide repeating units. Unlike xanthan gum, polyacrylamide has a more linear backbone, with functional groups attached along the chain. These groups enable the polymer to dissolve in water and expand within the solution, increasing resistance to flow. In some forms, partial substitution of functional groups introduces charges that further enhance interaction with aqueous solutions [4, p. 14961-14971].
Both xanthan gum and polyacrylamide are examples of polymers whose molecular architecture directly influences their macroscopic behavior. Despite differences in origin and structure, their long-chain nature and functional groups allow them to increase solution viscosity and control fluid mobility, making them valuable materials in applications such as polymer flooding and other fluid modification processes
The mechanisms of polymer flooding in harsh carbonate reservoirs
Previous research has identified several mechanisms by which polymer-based chemical enhanced oil recovery improves oil production. Among these mechanisms, the most dominant effect is the increase in viscosity of the injected aqueous phase caused by the presence of polymers. When polymers are dissolved in the injection fluid, the flow resistance of the displacing phase rises, which reduces its mobility relative to the oil.
This increase in viscosity allows the injected fluid to move more uniformly through the porous reservoir rock, preventing rapid flow through high-permeability channels. As a result, the injected polymer solution is able to displace hydrocarbons more effectively, contacting a larger portion of the reservoir and mobilizing oil that would otherwise remain trapped. In this way, polymer addition enhances macroscopic sweep efficiency and improves overall oil recovery.
Although other mechanisms such as adsorption, retention, and viscoelastic effects may contribute to recovery improvement, viscosity enhancement remains the primary and most widely recognized mechanism responsible for the effectiveness of polymer-based Chemical Enhanced Oil Recovery chemical EOR processes [1, p. 53; 7, p. 6087]. Contrasting water, this highly viscous polymer solution extents the oil in the porous medium and increases oil production.
In polymer flooding, the flow behavior of the aqueous phase is modified through an increase in brine viscosity and a corresponding change in its mobility (λW) [3]. At the same time, polymer presence leads to a reduction in the effective permeability of water (Kw). The combined influence of these effects is represented by the mobility ratio (M), which characterizes the relative movement of the injected fluid and oil within the porous medium [1, p. 53; 2, p. 550].
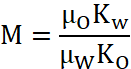 , (1)
, (1)
In these equation, the symbols λW and λO represent the flow capacity (mobility) of the brine and the oil phase (crude oil diluted with neutral kerosene), respectively. The terms KW of 10 mD and kO of proposed values ranging from 1.0 mPa.s to 23.5 mPa.s are the phase-specific effective permeability, while µW and µO correspond to the viscosities of the brine and oil. However, having a better understanding of concentration of the four major brine types according to Mg2+/Li+ mass ratio is so vita when determining brine mobility ratio [6, p. 29]. The mobility ratio expresses the relative ease at which brine solution displaces the oil within a porous structure. When the mobility ratio remains below unity, the displacement process tends to be uniform, allowing the injected fluid to efficiently sweep the reservoir. Conversely, mobility ratios greater than one reflect unfavorable flow conditions, under which the injected fluid advances too rapidly and bypasses significant portions of oil. Under such circumstances, polymer flooding becomes an effective approach, as increasing the viscosity of the injected brine through polymer addition reduces fluid mobility and enhances overall sweep efficiency [2, p. 550; 8; 9, p. 126-163].
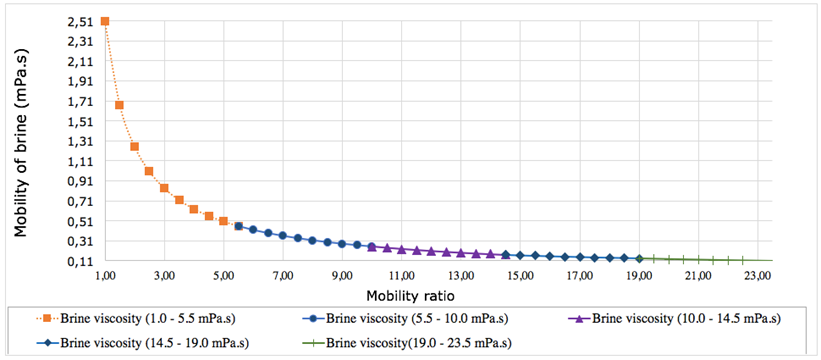
Fig. 1. The dependence of mobility brine on mobility ratio
The viscosity of polymer solution in brine
According to Hassan A.M and others [1, p. 53], the viscosity of polymer solution in brine is determined according to the equation:
![]() , (2)
, (2)
Where µP is the viscosity of polymer solution, CP is the concentration of polymer which ranges from 200 ppm to 3000 ppm, a of value 0.3 and b of value 0.7 are the constants which depends on the salinity and the type of polymer [5].
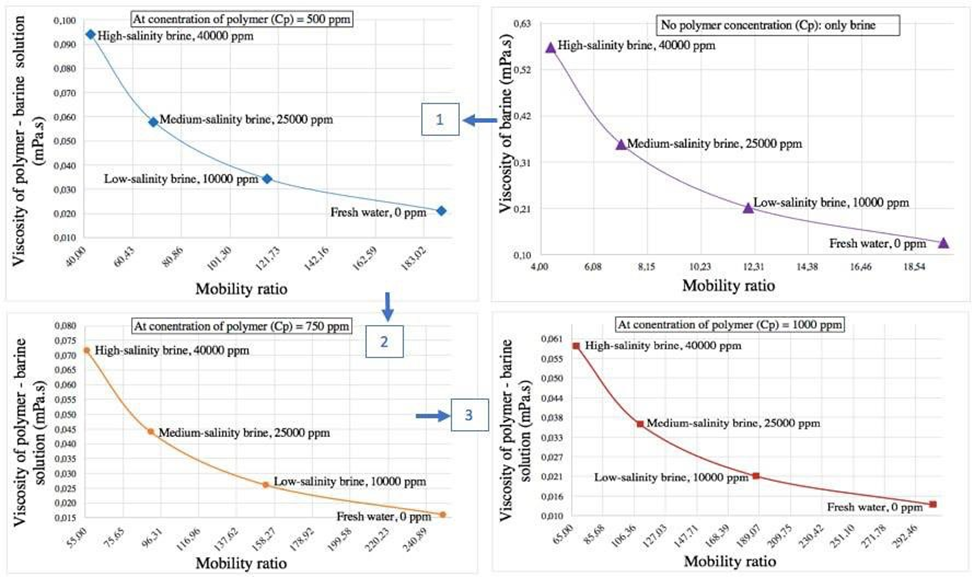
Fig. 2. The dependence of viscosity of polymer-brine solution in brine on mobility ratio
Conclusions
This study reveals how polymer-brine interaction plays a big positive impact in improving the flow properties of displacing fluids in carbonate reservoirs. Polymer regulates brine viscosity, hence improving the mobility of the displaced fluid and which subsequently boost sweep efficiency. According to the analysis, adding polymer solution from 500 ppm to 3000 ppm significantly regulated viscosity of the displacing fluid, depending on the brine’s salinity and makeup. With effective water permeability around 10 mD (Kw) and oil viscosity between 1–23.5 mPa·s (μo), the best polymer-brine combos stopped early breakthrough and ensured even oil displacement. Overall, picking the right polymer type, dose, and brine mix is key to getting the most oil out in tough, salty reservoir settings.
.png&w=384&q=75)
.png&w=640&q=75)